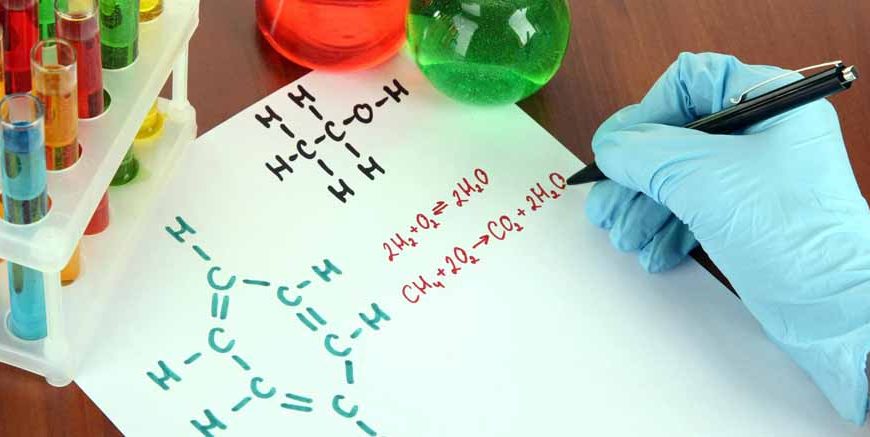Have you ever brushed your teeth early in the morning, or jumped into a cool swimming pool on a very hot summer afternoon? If you have done these simple things, you already know a little bit about a highly special group of chemicals. In science classes, teachers call them by a specific name. We call them the halogen family.
If you ask about the true halogen meaning, it comes from two very old Greek words. “Hal” means salt, and “gen” means to make or produce. So, the simple and exact meaning is “salt maker.” When these special chemicals mix with different metals, they create many different types of salts, just like the plain white salt you sprinkle on your french fries and popcorn!
Today, we are going to look closely at these amazing chemicals. We will find out where they live, how they behave, and why we need them in our daily lives.
Finding Their Home: The Science Map
To find these chemicals, we have to look at a large science map. This map is called the periodic table. It organizes all the known elements in the universe. If you want to find the halogens periodic table location, you have to look all the way to the right side of the map.
A very common question in a chemistry test is: halogens belong to which group? They live exactly in Group 17. Because they all live in the exact same column from top to bottom, they act just like a big family. If someone asks you if halogens belong to group in the modern periodic table, you can confidently say yes, they proudly make up the 17th column.
The halogen group periodic table section is very famous for a fun reason. It is the only group on the entire map that has elements in all three states of matter—gas, liquid, and solid—at a normal room temperature. This makes them incredibly unique to study.
Read More – How do you make science interesting for kids?
Meet the Family: Names and Faces
Let us meet the individual members of this chemical family. If a teacher asks for the name of halogens, you need to remember five main chemicals. They are Fluorine, Chlorine, Bromine, Iodine, and Astatine.
Fluorine:
This is the lightest member of the family. It is a pale yellow gas. It is also the most active and eager chemical in the entire periodic table. It loves to mix and react with almost everything it touches.
Chlorine:
This is a greenish-yellow gas. It has a very strong, sharp smell. If you have ever smelled bleach or freshly cleaned pool water, you know exactly what chlorine smells like.
Bromine:
This one is very unique. It is a heavy, dark red liquid. It is actually one of the only two elements on the entire periodic table that stays a liquid at room temperature. It gives off a strong, red vapor.
Iodine:
This is a dark purple or black solid rock. But it has a magic trick. If you heat it up, it does not melt into a liquid first. It turns straight from a solid rock into a beautiful, thick purple gas!
Astatine:
This is the heaviest and rarest member of the family. It is highly radioactive, and it is very hard to find in nature because it breaks down very quickly.
Read More – What Is Chlorine?
How Do They Behave?
Now let us talk about how they act. Understanding halogens properties is very easy if you think about them as kids who really want a specific toy to complete a set.
Every chemical element wants to have a full outer shell of tiny particles called electrons. Halogens have exactly seven electrons in their outer shell, but they need eight to be perfectly happy and stable. Because they are missing just one single piece to complete their set, they are extremely eager to grab that piece from anyone else.
This makes them highly reactive. They do not like to sit alone. In nature, you will almost never find a single atom of these elements just floating by itself. They are always holding hands with another element to share that missing piece.
Another interesting property is how they change as you go down the column. As you move from Fluorine at the top down to Iodine at the bottom, they get heavier, their colors get much darker, and they become a little bit slower to react with other things.
Everyday Uses in Our Lives
Why do we care about these chemicals? Because they help us stay safe and healthy every single day! Here are a few clear examples of how we use them.
- Healthy Teeth: Remember brushing your teeth? Your toothpaste contains a tiny amount of fluoride, which comes directly from fluorine. It makes the hard, white shell of your teeth very strong and stops painful cavities from forming.
- Clean Drinking Water: Chlorine is a wonderful cleaner. Cities put tiny, safe amounts of it in our drinking water and swimming pools. It actively hunts down and destroys bad germs and bacteria, keeping us safe from sickness.
- Healing Wounds: If you ever fall off your bicycle and get a bad cut on your knee, a doctor or your parents might clean it with a dark brown liquid. That liquid contains iodine. It perfectly cleans the open wound and stops dangerous infections.
- Bright Lights: Have you ever seen a very bright car headlight or a bright lamp in a store? We often call them halogen lamps. They use a tiny bit of iodine or bromine gas inside the glass bulb to make the light burn much brighter and last much longer than a normal, old-fashioned bulb.
Conclusion
To summarize, this special group of chemicals located in Group 17 is incredibly important to human life. From the pale yellow gas of fluorine to the dark purple solid rock of iodine, each member of this salt-making family has its own unique job. They are highly reactive because they are constantly searching for one final tiny piece to complete their outer shell.
As we close our chemistry books today, it leaves us with a highly thought-provoking idea. By themselves, pure halogens can be quite dangerous. Pure chlorine gas is toxic and not safe to breathe at all. Soft sodium metal explodes if it touches water. But when these two dangerous things meet and share their pieces, they instantly calm down and create Sodium Chloride. That is just plain table salt, something we safely eat every single day. It shows us that in science, combining two completely different, dangerous things can create something totally new, safe, and beautifully useful. To read more fun and educational articles, check out the EuroKids Blog, and visit our website for details on EuroKids Preschool Admission.
FAQs
Why are they called salt makers?
They are called salt makers because when they mix with metals (like sodium or potassium), they instantly create different types of natural salts.
Are these chemicals safe to touch?
In their pure forms, they are very strong and not safe to touch or breathe. However, when they mix with other elements to form compounds (like the fluoride in your toothpaste), they become completely safe and helpful.
Why is Bromine so special?
Bromine is special because it is a liquid at room temperature. Almost all other elements on the periodic table are either solid rocks or invisible gases.
Can I find these elements floating alone in nature?
No. Because they are so eager to react and find their missing electron, they are always found mixed and attached to other elements in nature.