Zinc is an important chemical element that we come across both in science textbooks and in everyday life. From protecting iron from rust to being an essential nutrient for human health, zinc plays a vital role in modern society. In Indian school chemistry syllabi, zinc is taught as part of metals and non-metals, the periodic table, and introductory chemistry chapters.
Here, we explain in detail what is zinc, what is its definition, meaning, atomic number, properties, chemistry, and uses, clearly and easily for students.
What Is Zinc?
Define Zinc
Zinc definition:
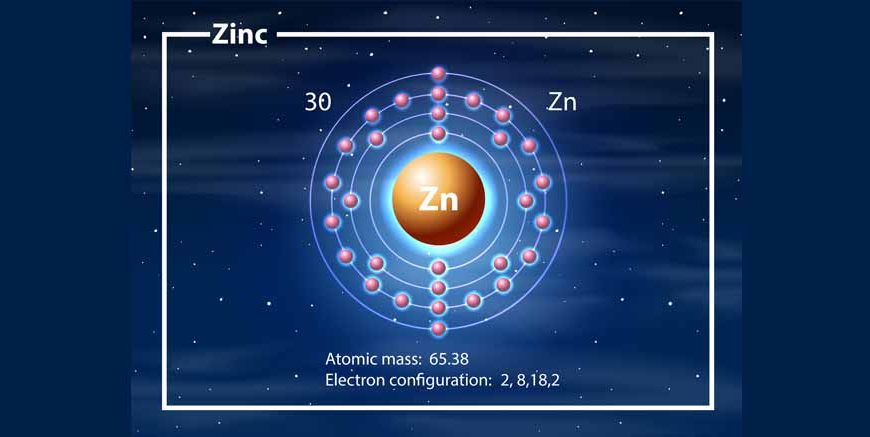
Zinc is a chemical element represented by the symbol Zn, having atomic number 30, and is classified as a metal.
In simple words, zinc is a bluish-white metal used widely in industries, medicine, construction, and daily household items.
Read More – Top Zinc-Rich Foods for Kids
Zinc Meaning
Zinc Meaning in English
The zinc meaning in English refers to a metallic element that resists corrosion and is used to coat iron and steel.
In India, zinc is commonly known as “जस्ता” (Jasta) in Hindi.
So, if you are wondering “jasta kya hai”, it means zinc metal.
Zinc Element and Its Discovery
The zinc element has been known to humans for thousands of years. Ancient Indians used zinc compounds in medicine and metallurgy, even before zinc was officially recognised as an element in modern chemistry.
Today, zinc is a well-defined element in the periodic table and is studied in detail in Zn chemistry.
Symbol for Zinc
- Symbol for zinc: Zn
- Zinc short form: Zn
What Is the Symbol of Zinc?
The symbol Zn comes from the German word Zink.
So, when students ask “what is Zn?”, the answer is: Zn is the chemical symbol of zinc.
Zn Full Form in Chemistry
There is no long expansion of Zn.
Zn full form in chemistry simply refers to the chemical symbol derived from its historical name.
Zinc in the Periodic Table
Zn Periodic Table Position
- Element name: Zinc
- Symbol: Zn
- Atomic number: 30
- Group: 12
- Period: 4
Both the zinc periodic table and the Zn periodic table refer to this exact placement.
So, if asked “what is the atomic number of zinc?”, the correct answer is 30.
This means zinc has:
- 30 protons
- 30 electrons
Zinc Atomic Structure
The zinc atomic structure shows how electrons are arranged.
Electron Configuration of Zinc
- 2, 8, 18, 2
This stable structure explains why zinc is less reactive than other metals.
Is Zinc a Metal or a Non-Metal?
A very common exam question is:
Is Zinc a Metal or Nonmetal?
- Zinc is a metal
Zinc belongs to the category of transition metals.
Read More – How do you make science interesting for kids?
Physical Appearance of Zinc
The physical appearance of zinc is:
- Bluish-white in colour
- Shiny when freshly cut
- Becomes dull on exposure to air
This appearance helps students identify zinc metal in laboratories.
Properties of Zinc
Physical Properties of Zinc
Some essential properties of zinc are:
- Solid at room temperature
- Brittle at normal temperature
- Becomes malleable when heated
- Good conductor of electricity
Density of Zn
- Density of Zn: approximately 7.14 g/cm³
Zinc Boiling Point and Melting Point
- Zinc melting point: ~419°C
- Zinc boiling point: ~907°C
These values are essential for Zn chemistry and industrial applications.
Zinc Chemical Name and Nature
- Zinc chemical name: Zinc
- Zn element name: Zinc
Zinc is a moderately reactive metal that forms compounds like zinc oxide and zinc sulphate.
Zinc Chemistry (Zn Chem)
Zinc chemistry focuses on how zinc reacts with:
- Acids (produce hydrogen gas)
- Oxygen (forms zinc oxide)
- Alkalis (form zincates)
In school-level Zn chem, zinc is often used to demonstrate displacement reactions.
Zinc Material and Zinc Metal
Zinc material is used mainly for protective purposes.
Zinc metal is rarely used alone but is combined with other metals.
About Zinc Metal
- Used as a coating
- Prevents rusting
- Increases the durability of iron
Uses of Zinc
Uses of Zinc in Daily Life
The uses of zinc are wide and practical:
1. Galvanisation
- Coating iron with zinc
- Prevents rusting
- Common in roofing sheets and pipes
2. Batteries
- Used in dry cells
- Zinc-carbon batteries
3. Alloys
- Brass (zinc + copper)
- Used in utensils and musical instruments
4. Medicine
- Zinc tablets for immunity
- Zinc oxide for skin creams
5. Coins and Hardware
- Locks, screws, handles
These are key zinc metal uses taught in Indian classrooms.
Uses of Zinc in India
In India, zinc is used in:
- Construction industry
- Electrical goods
- Ayurveda and modern medicine
- Agriculture (zinc sulphate for soil)
India is also a major producer of zinc globally.
Read More – National Science Day Activities & Experiments for Kids
About Zinc Element in Nutrition
Zinc is an essential micronutrient for humans.
It helps in:
- Growth
- Immunity
- Wound healing
However, this article focuses primarily on zinc as a chemical element, not on nutrition.
Zinc Meaning for Students
To simplify:
- Zinc meaning: A metal element used for protection and strength
- Zinc definition: A chemical element with symbol Zn and atomic number 30
How EuroKids Builds Strong EVS Foundations
When Environmental Studies (EVS) is introduced through observation, storytelling, nature walks, and hands-on exploration, young learners begin to understand their surroundings in a meaningful and lasting way. Instead of memorising facts about plants, animals, or seasons, children learn by experiencing and questioning the world around them. This is why structured early education programs focus on experiential and inquiry-based learning rather than rote memorisation.
At EuroKids, foundational EVS concepts are nurtured through the HEUREKA – Visible Thinking Curriculum. This approach encourages children to explore themes such as “My Family,” “My Community,” “Plants and Animals,” “Seasons and Weather,” and “Good Habits” through interactive discussions, thematic projects, role play, outdoor exploration, and guided observation.
The Visible Thinking framework helps children observe carefully, think deeply, and express their ideas confidently. Through activities like nature-based learning, environmental awareness projects, sensory exploration, and group conversations, children develop curiosity about how things grow, how communities function, and how they can care for the environment.
With a child-centric and play-based methodology refined over 20+ years and implemented across 1600+ preschools, EuroKids focuses on building awareness, responsibility, and curiosity first, allowing knowledge, empathy, and critical thinking skills to develop naturally over time.
About Zinc: Quick Summary
Here is a quick recap about zinc:
- Symbol: Zn
- Atomic number: 30
- Metal or nonmetal: Metal
- Colour: Bluish-white
- Uses: Galvanisation, batteries, alloys
- Hindi name: Jasta
Frequently Asked Questions (FAQs)
What Is Zn?
Zn is the chemical symbol for zinc.
What Is the Atomic Number of Zinc?
The atomic number of zinc is 30.
Is Zinc a Metal?
Yes, zinc is a metal.
What Is Jasta?
Jasta is the Hindi name for zinc.
What Are the Uses of Zinc?
Zinc is used to prevent rust, make alloys, batteries, and medicines.
Conclusion
Zinc is a versatile and essential element that plays a vital role in science, industry, and daily life. From its position in the periodic table to its wide uses in zinc metal applications, understanding zinc helps students build a strong foundation in chemistry. With properties like corrosion resistance and moderate reactivity, zinc stands out as a valuable metal in both textbooks and real life.
Learning about zinc not only prepares students for exams but also helps them understand how chemistry connects with everyday experiences.
To explore more learning ideas, simple explanations, and early childhood resources, parents
can visit the EuroKids Blog. Those who wish to understand the preschool journey better or
explore enrolment options can find complete information on the EuroKids Preschool Admission
Page, where learning is designed to be joyful, engaging, and child-friendly.